Preeminent Vascular Surgery Clinic in Hyderabad
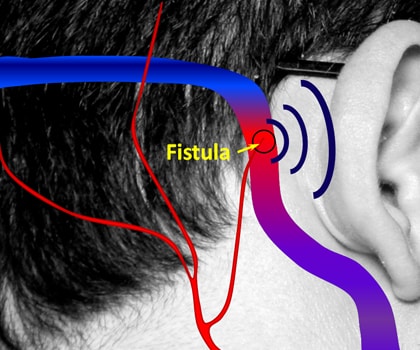
A Dural Arteriovenous Fistula (dAVF) is a condition that creates shunts from Dural vessels. They make about 10 – 15% of all AV shunting cerebral vascular malformations. dAVF is a condition of having peculiar connections between a vein and an artery over the spinal cord or the brain.
dAVF involves large brain veins, that usually develop due to blockage of one of the brain’s venous sinuses, which en routes the circulated blood from the brain back to the heart. These are typically passed on genetically.
Upper urinary tract obstruction, an infection caused by hydronephrosis, safety concerns (injury), and renal failure secondary to ureteral obstruction.
Dural Arteriovenous Fistula causing stunts from Dural Vessels can be treated by the DJ stenting procedure. The DJ stenting could be carried out under spinal anesthesia and in women even under local anesthesia. Local anesthesia is feasible as there aren’t any pain nerves in the bladder and solely distention of the Kidney is agonizing. The cystoscopy is used for the procedure. First, a guidewire is gone through the ureter to the Kidney. The DJ stent is then passed over the guidewire and pushed in via a pusher.
Usually, 6 weeks of placement of the DJ stent should suffice. Since it’s a remote body it mustn’t be kept for more than 3 months. It ought to be removed before that at any cost. If the stents aren’t removed for a long time, it may sentence to death. Stones can get deposited over the stent if there is insufficient water intake and it causes encrustation. If they are needed for a long time, stents must be replaced once in two months.
Q: Does the stent ever fall out?
A: Yes, uncommonly. If you notice any causality, then consult the doctor.
Q: When is a stent used?
A: A stent is placed if your Urologist thinks that urine might not drain well through the ureter.
